The vivo V30 5G and vivo V30 Pro 5G came out last month and now the company appears to be adding an SE variant. Its sub-brand could also be gearing up for the launch of a new tablet called the iQOO Pad 2 Pro, though a rebrand from the vivo Pad 3 Pro.
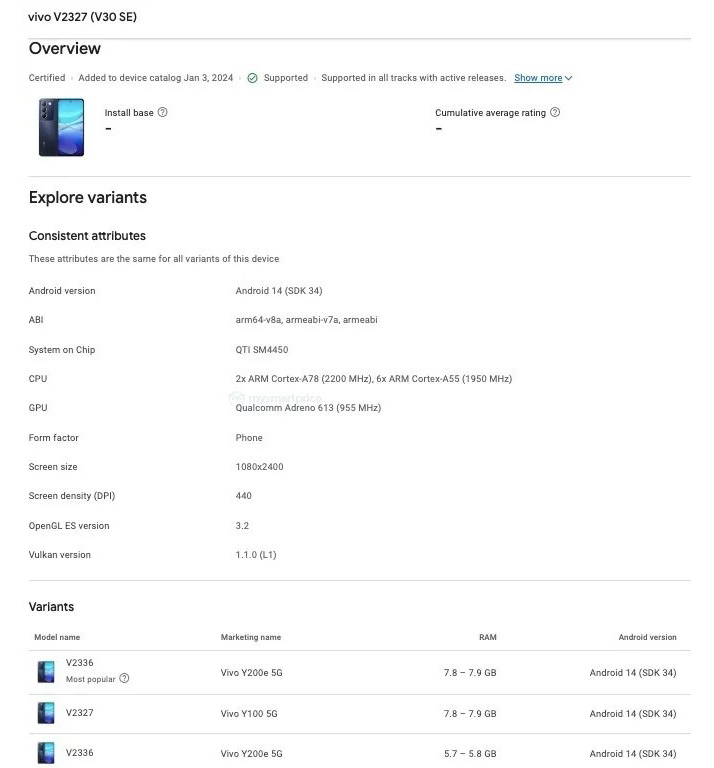
The new addition to the V30 Series, simply dubbed as the vivo V30 SE, surfaced on Google Play Console with the model number “V2327”. Specifications are meager in the listing, but the “Variants” tab hints that the V30 SE could be a rebranded version of the vivo Y200e 5G and/or the Y100 5G.

Thus, we can expect the vivo V30 SE to be rocking similar features: Snapdragon 4 Gen 2, a 6.67-inch Full HD+ AMOLED display with 120Hz refresh, dual rear cameras with 50-megapixel main shooter, and 5000mAh battery paired with 44W wired charging.
In fact, the Google Play Console listing confirms the phone’s CPU cores info (Cortex-A78 at 2.2GHz) that match those inside its variants. Android 14 is also confirmed for this new model, undoubtedly skinned with Funtouch OS 14.
As for the iQOO Pad 2 Pro (model number “DPD2329”), as mentioned, it could be a rebrand from the Chinese release vivo Pad 3 Pro 13. If that is indeed the case, then we could be seeing it equipped with a flagship Dimensity 9300 chipset, 13-inch LCD panel, and up to 16GB RAM plus 512GB config. As well as 11,500mAh battery with 66W wired charging.








0 Comments
Leave a Reply